- Modes of Operation: Adult/Pediatric, Passive (external ventilation), Spontaneous (simulated patient breathing).
- Functional Residual Capacity (FRC): Adjustable from 10 to 4000 mL to set baseline lung volume.
- Spontaneous Breath Rate: 0 to 100 breaths/min (supports high-frequency breathing scenarios).
- Muscle Pressure: 0-100 mbar to simulate patient inspiratory and expiratory effort.
- Airway Resistance: 5 to 500 mbar/L/s, configurable as linear or nonlinear (parabolic) resistance profiles.
- Lung Compliance: 3 to 300 mL/mbar, allowing simulation from very stiff to very compliant lungs.
- Leak: 0% to 100% (RP); simulate anything from a perfect seal to a completely open airway.
alveo Key Features
- Universal Compatibility: Seamlessly connect to all mechanical ventilators, anesthesia devices, and resuscitation bags, offering versatile training setups.
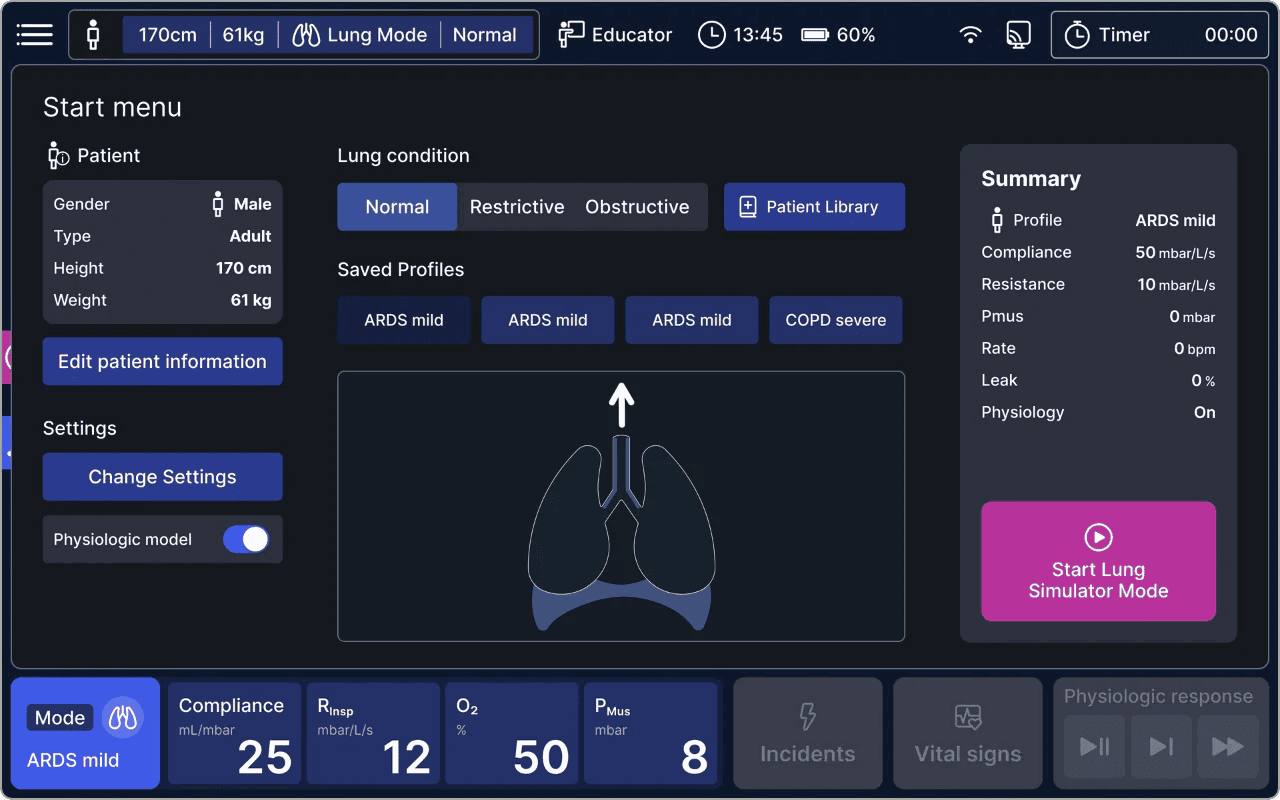
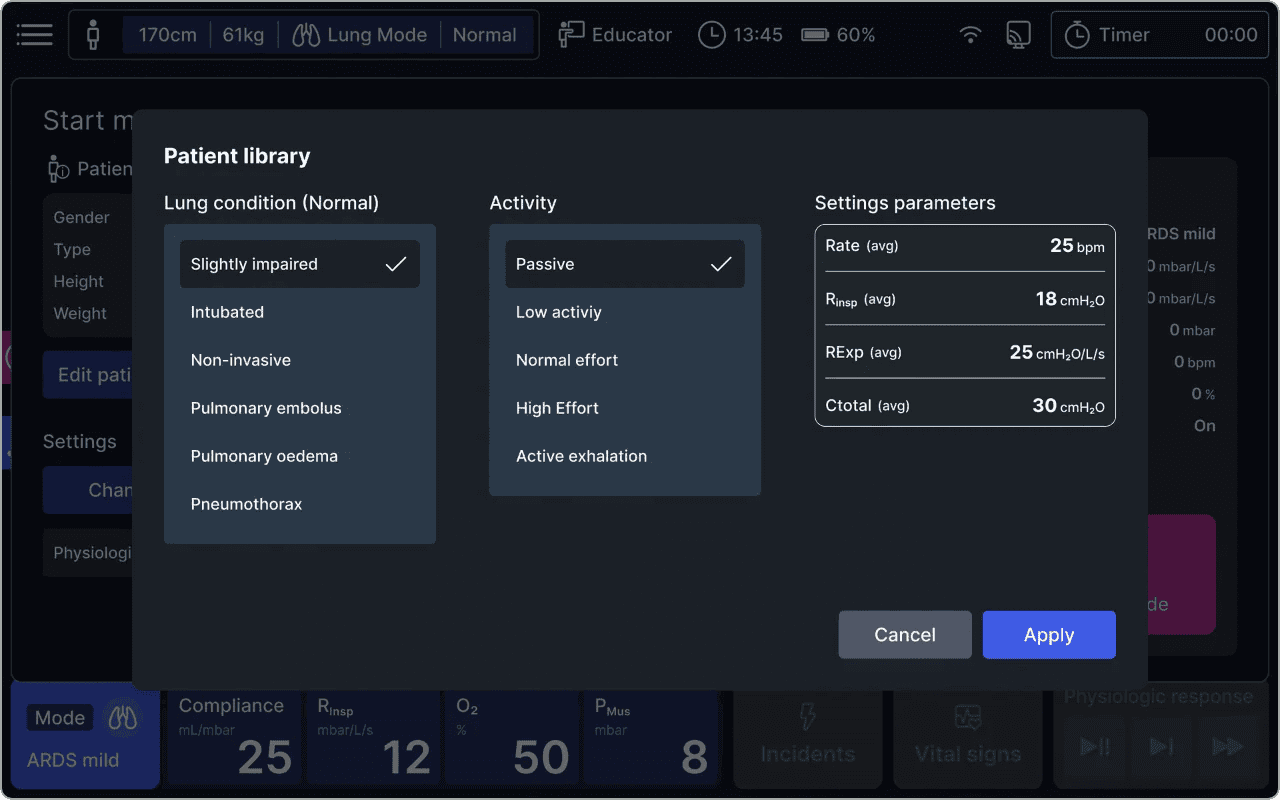
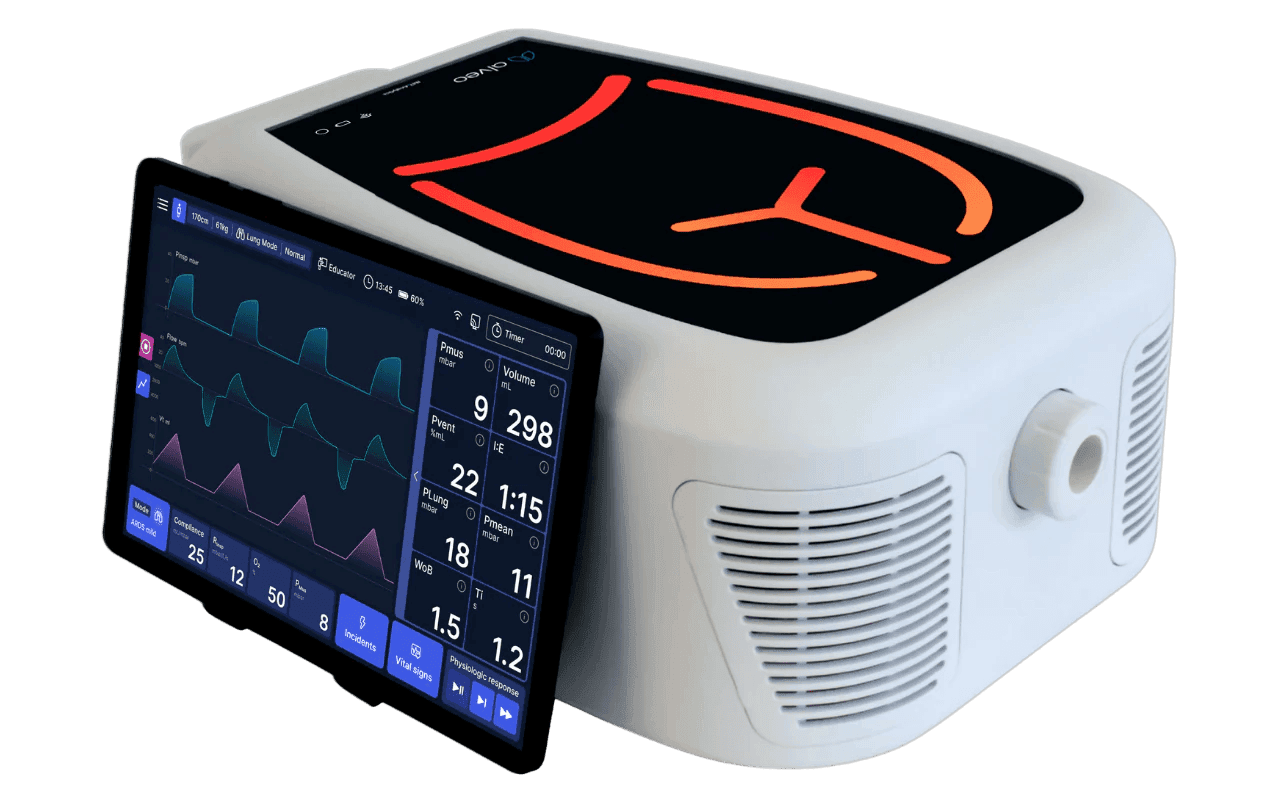
- Intuitive User Interface: alveo offers unparalleled ease of use, allowing users to control simulations effectively and enabling real-time observation of lung response. Navigation is straightforward, featuring quick menus for patient profiles and lung parameter adjustments.
- Advanced Physiological Modeling: alveo features a high-fidelity lung model that accurately simulates compliance and resistance, offering realistic breathing responses and physiological feedback. It emulates lung tissue properties and has a library of normal, restrictive, and obstructive lung conditions (e.g., ARDS, COPD) for comprehensive scenario training.
- Scripting and Automation: alveo supports remote control via the RS-232 interface, allowing advanced users to create custom breathing profiles and integrate with automated systems.
- Wireless Connectivity: Use alveo in wireless mode for monitoring and control, allowing you to comfortably use the tablets in simulation labs and research setups.
- Portable and Compact: alveo weighs only 6.8 kg and has compact dimensions, making it easy to transport between labs, classrooms, and manufacturing settings.
- alveo was developed in accordance with the ISO 13485 quality management standard and meets the requirements of IEC 61010.
- alveo guarantees reliability for accuracy and lung parameter adjustments through calibration in accordance with the ISO 17025 quality management standard.
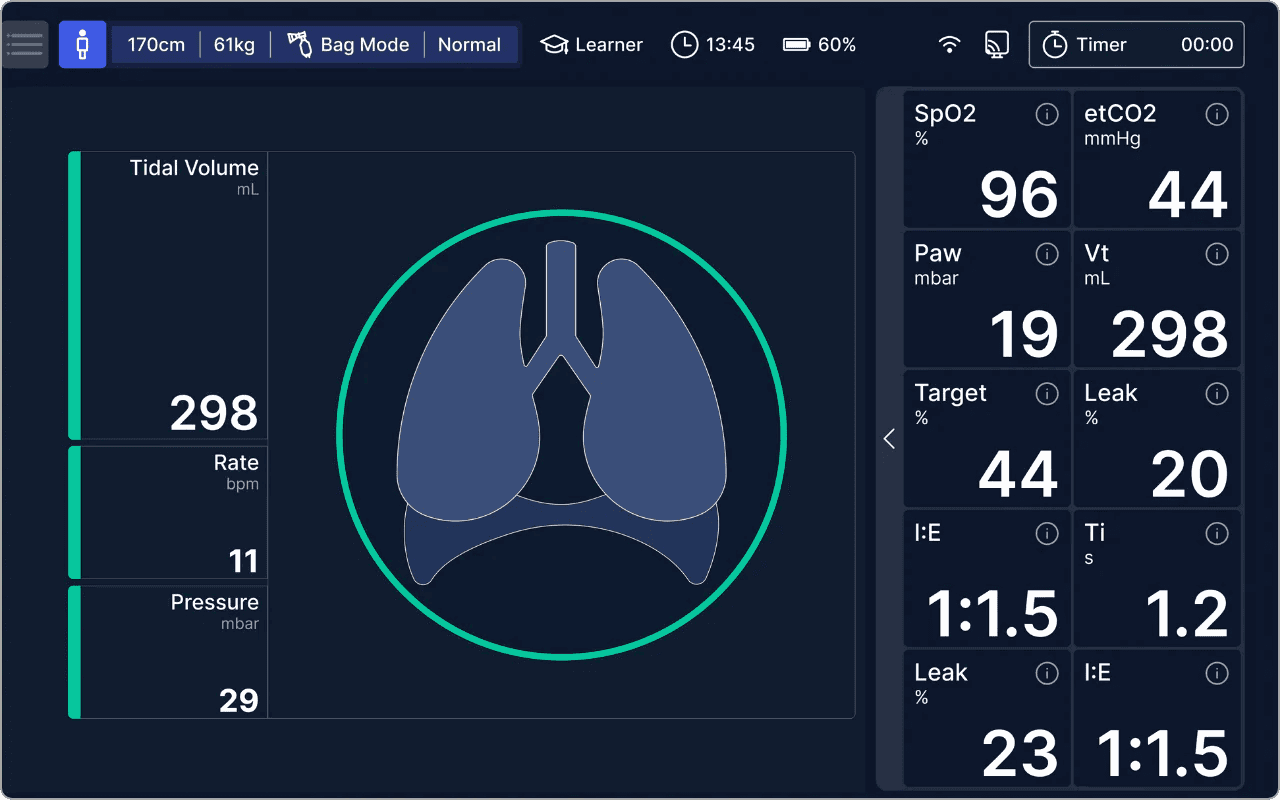
Realistic Lung Physiology Simulation
alveo sets a new standard in realism by simulating human lung behavior under various conditions. It utilizes a precision turbine-driven system to control airflow and lung compliance in real-time. This allows for quick adaptation to changes and spontaneous patient efforts, while also accounting for the viscoelastic properties of lung tissue to simulate stress responses, including gradual recruitment and hysteresis in pressure-volume curves.
alveo can reproduce a wide range of respiratory scenarios, from normal breathing to critical illness. Users can easily adjust settings to simulate conditions such as ARDS, COPD, or Asthma.
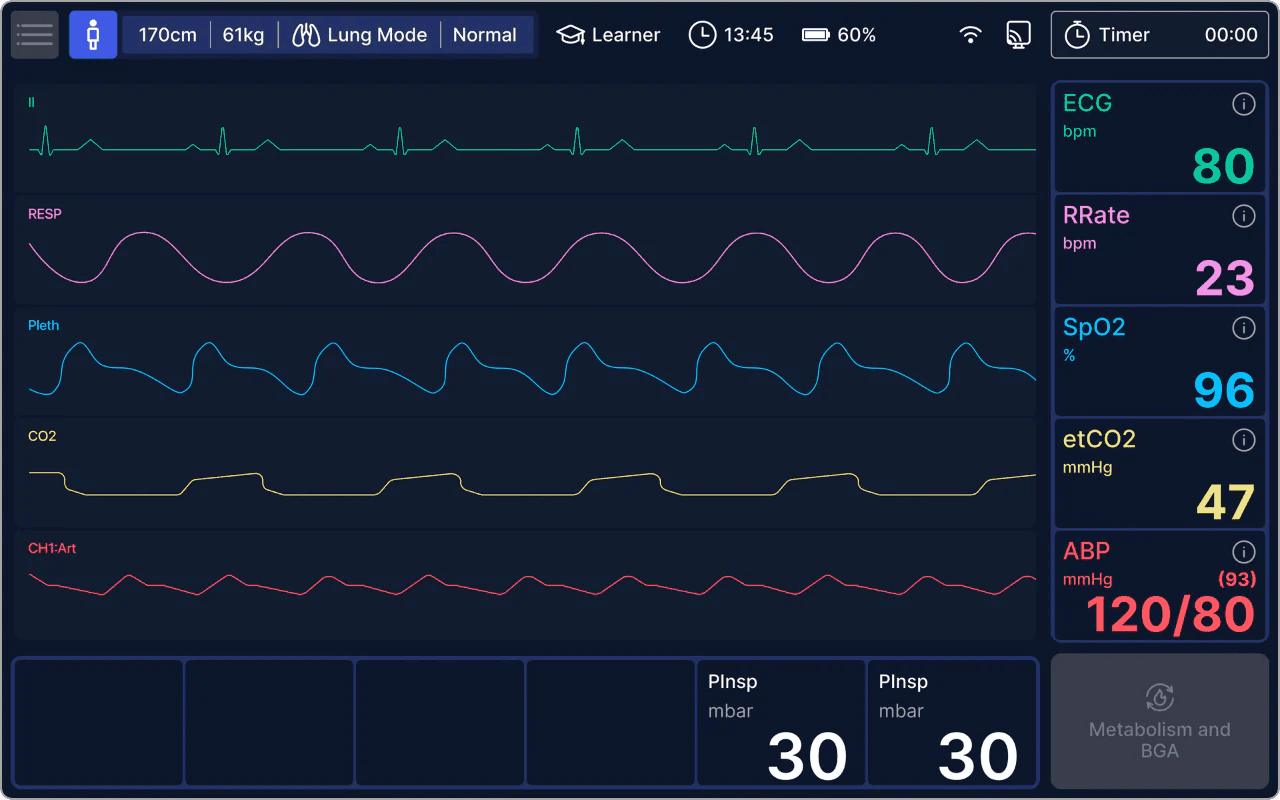
Use Cases: Clinical, Technical & Research
alveo is a versatile tool across various applications in clinical education, device engineering, and research innovation.
- Clinical Training and Education
alveo simulates realistic lung responses, enabling healthcare professionals to train in the management of ventilators and respiratory care. Trainees can practice adjusting settings, managing incidents, and enhancing their critical thinking and decision-making skills. - Development & Testing
alveo serves as a high-precision test lung for ventilator developers, enabling them to automate tests and verify performance with high-resolution data for enhanced safety and effectiveness. - Research & Innovation
alveo can help support research, advancing scientific knowledge in respiratory care.
Key Performance Specifications:
| CATEGORY | PARAMETER | RANGE | ACCURACY |
|---|---|---|---|
| Monitoring Parameters | |||
| Flow | Leak Flow (FlowLeak) | -150 to 300 L/min | ±3.7%* or ±0.2 L/min** |
| Peak Flow Inspiratory (FlowInsp) | -150 to 300 L/min | ±3.7%* or ±0.2 L/min** | |
| Peak Flow Expiratory (FlowExp) | -150 to 300 L/min | ±3.7%* or ±0.2 L/min** | |
| Pressure | Airway Pressure (Paw) | -80 to 120 mbar | ±1.5%* or ±0.3 mbar** |
| Mean Airway Pressure (PMean) | -80 to 120 mbar | ±1.5%* or ±0.3 mbar** | |
| Lung Pressure (PLung) | -80 to 120 mbar | ±1.5%* or ±0.3 mbar** | |
| Mean Lung Pressure (PLungMean) | -80 to 120 mbar | ±1.5%* or ±0.3 mbar** | |
| Positive End-Expiratory Pressure (PEEP) | -80 to 120 mbar | ±1.5%* or ±0.3 mbar** | |
| Total PEEP (PEEP) | -80 to 120 mbar | ±1.5%* or ±0.3 mbar** | |
| Volume | In- & Expiratory Tidal Volume (Vti, Vte) | 0–10 L | ±3.7%* or ±0.2 L/min** |
| In- & Expiratory Minute Volume (Vi, Ve) | 0–10 L | ±3.7%* or ±0.2 L** | |
| End-Expiratory Lung Volume (EELV) | 0–10 L | ±3.7%* or ±0.2 L/min** | |
| Timing | Respiratory Rate (Rate) | 1–2000 bpm | ±1.0% or ±1 bpm** |
| Inspiratory Time (Ti) | 0.01–60 s | ±0.2 s | |
| Expiratory Time (Te) | 0.01–90 s | ±0.2 s | |
| I:E Ratio | 1:300–300:1 | ±2%* | |
| Ti/Tcyc | 0–100% | ±5%* | |
| Work | Total Work (Inspiratory) | 0–1000 J/L | ±5%* |
| Work of Breathing (Patient) | 0–1000 J/L | ±5%* | |
| Power | WTotal | 0–1000 J/min | ±5%* |
| Settings | |||
| Compliance (Linear / S-Curve) | Crs, CLung, Ccw | 3–300 mL/mbar Adult/Pediatric; 0.2–300 mL/mbar Neonatal | ±5%* |
| Resistance (Linear / Parabolic) | Rinsp, Rexp | 5–500 mbar/L/s | Parabolic: ±10%*; Linear: ±20%* |
| Spontaneous | Rate | 0–100 bpm | ±1.0% or ±1 bpm** |
| Muscular Effort Inspiratory (PMusInsp) | 0–100 mbar | ±3% or ±0.5 mbar** | |
| Muscular Effort Expiratory (PMusExp) | 0–100 mbar | ±3% or ±0.5 mbar** | |
| Neural Inspiratory Time (TMus) | 0.1–10 s | ±0.1 s | |
| Rise / Hold / Release Inspiratory | 0.1–30% / 0.1–90% / 0.1–30% | ||
| Rise / Hold / Release Expiratory | 0.1–30% / 0.1–90% / 0.1–30% | ||
| Variation Rate | 0–30% | ||
| Variation PMus | 0–30% | ||
| Additional Settings | Leak | Off, 0.1–100% | |
| Functional Residual Capacity (FRC) | 10–4000 mL | ||
| Recruitability | Good, Medium, Poor, None | ||
| Physiology Model | On / Off | ||
| Units | Flow | L/min, mL/min | |
| Pressure | mbar, cmH2O | ||
| Work | J/min, mJ/min, J/L | ||
| Temperature | °C, °F | ||
| Gas Types | Air, O2 | ||
| Gas Standards | STP, BTPS | ||
| Additional Monitoring | |||
| In Flow Channel | Oxygen | 0–100% | ±1% O2** |
| Temperature | 0–50 °C | ±2%** | |
| Humidity | 0–100% RH (non condensing) | ±5% RH** | |
| Atmospheric Pressure (PAtmo) | 700–1200 mbar | ±1.5%* or ±5 mbar** | |
| Waveforms | |||
| Pressure | Paw, PLung, Paw/PLung, PMus/Paw, PMus/PLung | ||
| Flow | Flowaw, FlowLeak, Flowaw/FlowLeak | ||
| Volume | Volume | ||
| Vital Signs (Calculated) | |||
| Vital Signs (Waveforms) | ECG, Plethysmography, Arterial Blood Pressure, End-Tidal CO2, Respiratory Rate | ||
| Vital Signs (Numeric) | Heart Rate, Respiratory Rate (RR), etCO2, SpO2, Arterial Blood Pressure | ||
| Metabolism | V̇O2, V̇CO2 | ||
| Respiratory | V̇d/Vt, V̇Q, Shunt, PaO2/FiO2 Index | ||
| Arterial Blood Gas | PaO2, PaCO2, AaDO2, pH, BE, Hb | ||
| Compliance | Crs, CLung, Ccw | ||
| Resistance | Rinsp, Rexp | ||
| General Information | |||
| Power | 100–240 VAC (±20%), 50–60 Hz, 2.0 A | ||
| Battery Operation | 3 hours | ||
| Weight | Device 6.8 kg/16.5 lbs, Tablet 0.48 kg/1.06 lbs | ||
| Dimensions (w × d × h) | 410 × 295 × 160 mm; 16.14 × 11.61 × 6.29 in | ||
| Data Storage & Retrieval | Via RS232 and external Tools | ||
| Sample Rate | 1 kHz | ||
| Display | 11" Android Tablet, 1920×1200 (WUXGA) | ||
| Interfaces | 2 × USB-C, Ethernet, CAN, COM1, COM2, RS232 | ||
| Calibration & Maintenance | Annually | ||
| Operating Temperature | 10–40 °C (50–104 °F) | ||
| Operating Humidity | 10–90% RH, non-condensing | ||
| Approvals | CE, CSA (North America), IEC 61010-1:2010, IEC 61326-2:2012 | ||
|
The greater tolerance is valid. * Tolerance related to the measured value. ** Absolute tolerance, with steady air flow. |
|||